
Apossible mechanism for the gas phase reaction of no and h2 is as follows: step 1 2no n2o2 step 2 n2o2 + h2 n2o + h2o step 3 n2o + h2 n2 + h2o which of the following statements concerning this mechanism is not directly supported by the information provided? a. n2o2 is an intermediate. b. all steps are bimolecular reactions. c. the rate expression for step 1 is rate = k[no]2. d. step 1 is the rate determining step. e. there is no catalyst in this reaction. 1 points question 9

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:30
How many moles of air are there in a human lung with a volume of 2.4 l at stp? explain your answer
Answers: 1

Chemistry, 22.06.2019 11:30
Which statement best describes the flow of energy in this scenario
Answers: 1

Chemistry, 22.06.2019 21:30
Athe top of a hill, an athlete on a skateboard has x joules of mechanical energy. how much mechanical energy will she have at the bottom of the hill? ignore the effects of friction.
Answers: 1

You know the right answer?
Apossible mechanism for the gas phase reaction of no and h2 is as follows: step 1 2no n2o2 step 2 n...
Questions

Social Studies, 10.09.2019 01:30


French, 10.09.2019 01:30

Mathematics, 10.09.2019 01:30

Mathematics, 10.09.2019 01:30

Mathematics, 10.09.2019 01:30

French, 10.09.2019 01:30

SAT, 10.09.2019 01:30

French, 10.09.2019 01:30


Mathematics, 10.09.2019 01:30

Mathematics, 10.09.2019 01:30

Mathematics, 10.09.2019 01:30


Social Studies, 10.09.2019 01:30



History, 10.09.2019 01:30


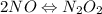 (fast)
(fast)![[NO]^{2}](/tpl/images/0174/7336/f0d3d.png)
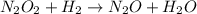
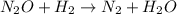 (fast)
(fast)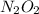 is intermediate in nature.
is intermediate in nature.

