

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:20
Describing intermolecular forces use the drop down menus to match the type of intermolecular force to its name dipole dipole interactions dipole induced dipole interactions london dispersion forces hydrogen bond van der waals forces done
Answers: 1

Chemistry, 22.06.2019 09:30
Melissa is interested in her family tree and how her family has changed over its many generations. melissa probably more closely resembles
Answers: 2

Chemistry, 22.06.2019 14:30
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4.0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3

Chemistry, 22.06.2019 19:00
What information does a complete ionic equation give that the balanced equation doesn’t show?
Answers: 1
You know the right answer?
Classify each of the following substances as either a strong or weak acid, strong or weak base, or a...
Questions


English, 22.12.2019 21:31

English, 22.12.2019 21:31

Mathematics, 22.12.2019 21:31

Mathematics, 22.12.2019 21:31


History, 22.12.2019 21:31

Geography, 22.12.2019 21:31


History, 22.12.2019 21:31


Health, 22.12.2019 21:31

Mathematics, 22.12.2019 21:31

Chemistry, 22.12.2019 21:31

History, 22.12.2019 21:31

Spanish, 22.12.2019 21:31

Social Studies, 22.12.2019 21:31


Mathematics, 22.12.2019 21:31

History, 22.12.2019 21:31

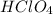 is a strong acid,
is a strong acid, 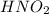 is a weak acid, NaBr is a soluble salt and
is a weak acid, NaBr is a soluble salt and 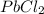 is an insoluble salt
is an insoluble salt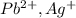 and Hg^{+}[/tex].
and Hg^{+}[/tex].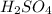 ,
, 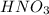 ,
, 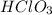 and
and 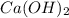 ,
, 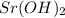 and
and 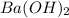 are strong bases.
are strong bases.

