Chemistry, 22.08.2019 16:30 gujacksongu6
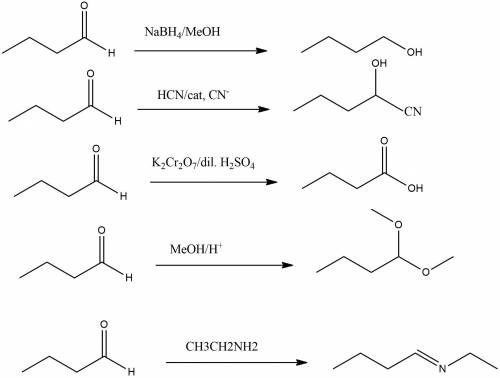
Give the products of the reaction of butanal, with: (a) sodium borohydride, in methanol (b) hydrogen cyanide, with catalytic cyanide (c) potassium dichromate in dilute sulfuric acid; (d) methanol, with a trace of acid catalyst; (e) ethylamine (ch3ch2nh2)

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 23:00
Will mark brainliest26. which of these statements are true? (3 points)a. gases are compressibleb. gases fill their containers completelyc. the pressure of a gas is independent of the temperatured. gases have masse. gases exert pressuref. the pressure of a gas is dependent on the volumeg. gas pressure results from the collisions between gas particlesh. gases have a definite volume and shape
Answers: 1

Chemistry, 22.06.2019 04:20
Which formula can be used to calculate the molar mass of ammonia (nh3)? molar mass of n + molar mass of h 3 × molar mass of n + molar mass of h molar mass of n + 3 × molar mass of h 3 × molar mass of n + 3 × molar mass of h
Answers: 1

Chemistry, 22.06.2019 08:00
An electron moved from shell n = 2 to shell n = 1. what most likely happened during the transition? a fraction of a photon was added. a photon of energy was absorbed. a fraction of a photon was removed. a photon of energy was released.
Answers: 1

You know the right answer?
Give the products of the reaction of butanal, with: (a) sodium borohydride, in methanol (b) hydroge...
Questions


Social Studies, 29.07.2019 18:30

English, 29.07.2019 18:30








English, 29.07.2019 18:30

Chemistry, 29.07.2019 18:30





Mathematics, 29.07.2019 18:30

Mathematics, 29.07.2019 18:30

Mathematics, 29.07.2019 18:30

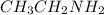 to produce imine.
to produce imine.