Chemistry, 22.08.2019 17:10 zoeyandblaze
Write the expression for the equilibrium constant kp for the following reaction. enclose pressures in parentheses and do not write the chemical formula as a subscript. for example, enter (pnh3)2 as (p nh3)2 .if either the numerator or denominator is 1, enter 1nh4hs(s) ↔ nh3(g) + h2s(g)

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 11:00
The number to the right of an element's symbol (ex. c-12) identifies the of an isotope.
Answers: 1



You know the right answer?
Write the expression for the equilibrium constant kp for the following reaction. enclose pressures i...
Questions



Mathematics, 30.11.2020 02:00

Social Studies, 30.11.2020 02:00

Mathematics, 30.11.2020 02:00

Mathematics, 30.11.2020 02:00





Mathematics, 30.11.2020 02:00

Mathematics, 30.11.2020 02:00



Mathematics, 30.11.2020 02:00

Advanced Placement (AP), 30.11.2020 02:00




Computers and Technology, 30.11.2020 02:00

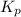 is written below.
is written below.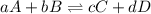
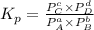
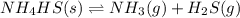
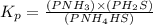
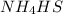 will be 1 because it is solid.
will be 1 because it is solid.