Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 12:00
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2


Chemistry, 22.06.2019 21:30
If you burn 46.6 g of hydrogen and produce 416 g of water, how much oxygen reacted
Answers: 3

Chemistry, 22.06.2019 22:30
What if it is did darwin used to support his theory of evolution
Answers: 1
You know the right answer?
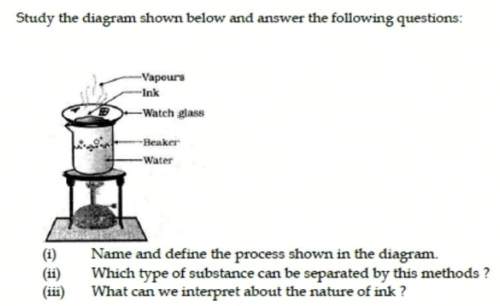
Study the diagram below. a) name the process. b) which type of substance can be separated. c)what ca...
Questions








Mathematics, 16.09.2019 20:40


Advanced Placement (AP), 16.09.2019 20:40

Social Studies, 16.09.2019 20:40

English, 16.09.2019 20:40

Physics, 16.09.2019 20:40


Biology, 16.09.2019 20:40


English, 16.09.2019 20:40

Mathematics, 16.09.2019 20:40

Biology, 16.09.2019 20:40

Mathematics, 16.09.2019 20:40