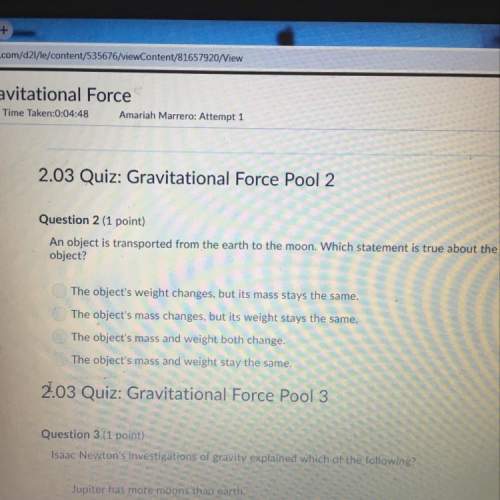
Acertain half-reaction has a standard reduction potential =e0red+0.13v . an engineer proposes using this half-reaction at the anode of a galvanic cell that must provide at least 1.10v of electrical power. the cell will operate under standard conditions. note for advanced students: assume the engineer requires this half-reaction to happen at the anode of the cell. is there a minimum standard reduction potential that the half-reaction used at the cathode of this cell can have? if so, check the "yes" box and calculate the minimum. round your answer to 2 decimal places. if there is no lower limit, check the "no" box. yes, there is a minimum. =e0red v no minimumis there a maximum standard reduction potential that the half-reaction used at the cathode of this cell can have? if so, check the "yes" box and calculate the maximum. round your answer to 2 decimal places. if there is no upper limit, check the "no" box. yes, there is a maximum. =e0red v no maximumby using the information in the aleks data tab, write a balanced equation describing a half reaction that could be used at the cathode of this cell. note: write the half reaction as it would actually occur at the cathode. e

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:30
Select the correct answer. when carbon dioxide dissolves in water, it sometimes reacts with water to form carbonic acid as in this balanced equation: co2 + h2o → h2co3. if 495 milliliters of carbon dioxide at 25°c and 101.3 kilopascals reacts with excess water, what is the theoretical yield of carbonic acid? use the periodic table and the ideal gas resource a. 0.889 g b. 1.10g c. 1.27g d. 2.02g what's the answer! quick!
Answers: 1

Chemistry, 22.06.2019 08:30
Which of the following would be an accurate picture of the earth during the summer time of the northern hemisphere?
Answers: 1

Chemistry, 22.06.2019 09:00
Identify the electromagnets with poles that are reversed from the electromagnet shown above
Answers: 3

You know the right answer?
Acertain half-reaction has a standard reduction potential =e0red+0.13v . an engineer proposes using...
Questions

Mathematics, 22.04.2020 22:48



Mathematics, 22.04.2020 22:48

History, 22.04.2020 22:48

Mathematics, 22.04.2020 22:48



Mathematics, 22.04.2020 22:48

Arts, 22.04.2020 22:48

English, 22.04.2020 22:48

History, 22.04.2020 22:49


Mathematics, 22.04.2020 22:49

Mathematics, 22.04.2020 22:49

Mathematics, 22.04.2020 22:49

History, 22.04.2020 22:49

Mathematics, 22.04.2020 22:49

Mathematics, 22.04.2020 22:49