
Chemistry, 27.08.2019 16:30 ashleyaparicio7116
Match. the expression of concentration that provides the moles of solute per kilograms of solvent is solution is made up of 0.15 grams of sodium chloride in 1 liter of water. for this solution, is water. a solution is made up of 0.15 grams of sodium chloride in 1 liter of water. for this solution, the sodium chloride. if you place 5 moles of sodium chloride and 4 moles of sucrose into 11 moles of water, the sodium chloride would be 0.25.a way to express concentration that provides the moles of solute per liter of solution is

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 01:00
Which of the following is always a reactant in a combustion reaction? oxygen nitrogen hydrogen carbon
Answers: 1

Chemistry, 22.06.2019 02:40
The difference between the atomic number of an element and the element’s atomic mass is the number of ions.
Answers: 3

Chemistry, 22.06.2019 05:00
If the density of water is 1.0 g/cm3, which of these materials would float in water, based on their densities? check all that apply. aluminum cork iron lead wax
Answers: 1

Chemistry, 22.06.2019 06:00
Why is permeable soil best for plants that need a lot of drainage?
Answers: 1
You know the right answer?
Match. the expression of concentration that provides the moles of solute per kilograms of solvent is...
Questions


Computers and Technology, 04.12.2020 05:00

Mathematics, 04.12.2020 05:00

Chemistry, 04.12.2020 05:00






Mathematics, 04.12.2020 05:00


Mathematics, 04.12.2020 05:00

Mathematics, 04.12.2020 05:00





English, 04.12.2020 05:00


Chemistry, 04.12.2020 05:00

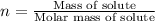
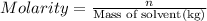
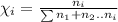
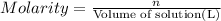
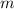 ) is defined as the ratio between the moles of the solute and the mass of the solvent in kilograms only:
) is defined as the ratio between the moles of the solute and the mass of the solvent in kilograms only:![m=\frac{n_{solute}}{m_{solvent}} [=]\frac{mol}{kg}](/tpl/images/0202/7993/d516f.png)
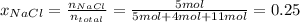
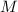 ) is defined as the ratio between the moles of the solute and the volume of the solution in liters only:
) is defined as the ratio between the moles of the solute and the volume of the solution in liters only:![M=\frac{n_{solute}}{V_{solution}} [=]\frac{mol}{L}](/tpl/images/0202/7993/a5282.png)


