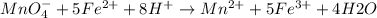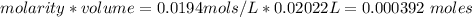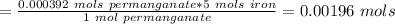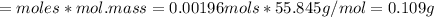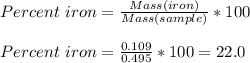Chemistry, 28.08.2019 05:20 kayleenfifep3hgi4
A0.495 g sample of iron containing salt required 20.22 ml of 0.0194 m permanganate solution to reach the endpointof a titration. what is the percent of iron (55.845 g/mol) in the salt

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 07:00
The blackbody curve for a star name zeta is shown below. what is the peak wavelength for this star ?
Answers: 1


Chemistry, 22.06.2019 13:00
These questions are based on the attached photo. the experiment is about burning magnesium metal with oxygen. 1. write the balanced chemical equation for the reaction you are performing. 2. calculate the mass of magnesium metal used in each trial. o trial 1: o trial 2: 3. calculate the actual yield of magnesium oxide for each trial. o trial 1: o trial 2: 4. magnesium is the limiting reactant in this experiment. calculate the theoretical yield of mgo for each trial. o trial 1: o trial 2: 5. determine the percent yield of mgo for your experiment for each trial. o trial 1: o trial 2: 6. determine the average percent yield of mgo for the two trials. your company currently uses a process with a similar cost of materials that has an average percent yield of 91 percent. if the average percent yield of this process is higher than that, this could save the company money. what is your recommendation to the company? support your recommendation using your data, calculations, and understanding of stoichiometry gathered from this lab.
Answers: 1

Chemistry, 23.06.2019 00:10
Apropane torch is lit inside a hot air balloon during preflight preparations to inflate the balloon. which condition of the gas remains constant
Answers: 2
You know the right answer?
A0.495 g sample of iron containing salt required 20.22 ml of 0.0194 m permanganate solution to reach...
Questions




Mathematics, 16.10.2020 05:01


Spanish, 16.10.2020 05:01

Social Studies, 16.10.2020 05:01

Mathematics, 16.10.2020 05:01

Mathematics, 16.10.2020 05:01


Mathematics, 16.10.2020 05:01

Social Studies, 16.10.2020 05:01




Mathematics, 16.10.2020 05:01

Physics, 16.10.2020 05:01


English, 16.10.2020 05:01

Spanish, 16.10.2020 05:01