
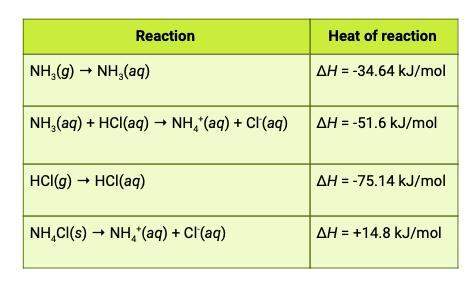
The heat of reaction for the process described in (a) can be determined by applying hess's law. the heats of reaction shown in the table below can be obtained experimentally or looked up in tables of enthalpy data. which two of these heats of reaction would be the easiest and safest to measure in the laboratory, and which two are better obtained through reference sources? why? hint: consider whether a reaction takes place in aqueous solution or instead involves noxious gases.
this is the equation for a: δh25° = ( δh25° nh3 + δh25° hcl) - ( δh25° nh4cl)
and attached is the table

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:00
Will give brainliest it is a lab from k12 here is the linkfor each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. type your answer here. (score for question 3: of 5 points) were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. type your answer here. (score for question 4: of 5 points) make a general statement about the reactivity of the metals in this experiment. type your answer here.
Answers: 2

Chemistry, 22.06.2019 10:40
If an area has high air pressure and low humidity, what type of weather will it most likely have? plz !
Answers: 1

Chemistry, 22.06.2019 12:10
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 14:50
How are evaporation and sublimation similar? a both involve the formation of a gas. b both release energy to the surroundings. c both take place throughout a solid. d both take place at the surface of a liquid.
Answers: 1
You know the right answer?
The heat of reaction for the process described in (a) can be determined by applying hess's law. the...
Questions





SAT, 18.03.2021 02:30

Advanced Placement (AP), 18.03.2021 02:30

Social Studies, 18.03.2021 02:30

Chemistry, 18.03.2021 02:30






Engineering, 18.03.2021 02:30

Mathematics, 18.03.2021 02:30

Mathematics, 18.03.2021 02:30

Mathematics, 18.03.2021 02:30



Mathematics, 18.03.2021 02:30



