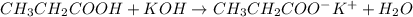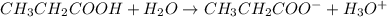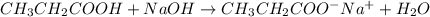write a balanced equation for the reaction of propanoic acid with each of the following:
a. k...

Chemistry, 30.08.2019 05:30 reneewilliams20
write a balanced equation for the reaction of propanoic acid with each of the following:
a. koh
b. h2o
c. naoh

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:30
In which layer of earth do most earthauakes occur a_ inner core b_outer core c_mantle d_crust
Answers: 1

Chemistry, 22.06.2019 21:30
Plzz a sample of table sugar (sucrose, c12h22o11) has a mass of 7.801 g. ● a) calculate the number of moles of c12h22o11 in the sample b) calculate the number of moles of each element in c12h22o11 (number of moles of c, number of moles of h & number of moles of o) in the sample. (use your answer from part a as your starting point.) show your work and highlight your final answer. calculate the number of atoms of each element in c12h22o11 (number of atoms of c, number of atoms of h & number of atoms of o) in the sample. (use your answers from part b as your starting for each element.) show your work and highlight your final answer.
Answers: 1

Chemistry, 22.06.2019 23:30
Rank the following four acids in order of increasing bronsted acidity : h2f+ , ch3oh, (ch3)2oh+ , ch3sh2+
Answers: 3

Chemistry, 23.06.2019 00:20
How many lone pairs of electrons are on the central atom of no3- and what is the molecular shape? one, trigonal planar zero, trigonal pyramidal zero, trigonal planar one, tetrahedral one, trigonal pyramidal
Answers: 1
You know the right answer?
Questions

Social Studies, 13.10.2020 20:01


History, 13.10.2020 20:01

Mathematics, 13.10.2020 20:01

Biology, 13.10.2020 20:01

Mathematics, 13.10.2020 20:01



English, 13.10.2020 20:01



Mathematics, 13.10.2020 20:01

Mathematics, 13.10.2020 20:01

Arts, 13.10.2020 20:01

Mathematics, 13.10.2020 20:01

Mathematics, 13.10.2020 20:01

Mathematics, 13.10.2020 20:01

Health, 13.10.2020 20:01


SAT, 13.10.2020 20:01