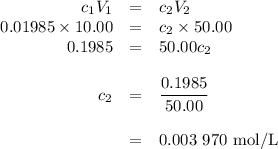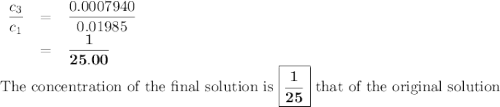Chemistry, 31.08.2019 03:10 dsaefong00
the molar concentration (m) of a solution prepared by dissolving 0.2362g of cr(no3)3 in a 50-ml volumetric flask is 0.01985m, where the molecular weight for cr(no3)3 = 238.01g/mol.
a. suppose you want to prepare another solution containing chromium nitrate that is 25 times less concentrated than the one prepared above. given a choice of 10-ml and 5-ml pipets and 50-ml and 100-ml volumetric flasks, explain how you would proceed in preparing the new diluted solution. in addition, calculate the concentration for the new diluted solution. show all work. your final value should have the correct unit and number of significant figures. hint: you will most likely need two dilution steps in order to obtain the desired concentration. note: you may not reuse the same pipet or combine different pipets within the same dilution step. you may reuse the pipet and/or volumetric flask in the different dilution step.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 17:40
If 3 moles of a compound use 24 j of energy in a reaction, what is the a hreaction in j/mol?
Answers: 1

Chemistry, 22.06.2019 20:30
Which states of matter have particles that move independently of one another with very little attraction?
Answers: 1

Chemistry, 22.06.2019 22:30
Calculate the concentration of all species in a 0.165 m solution of h2co3.
Answers: 1

Chemistry, 23.06.2019 00:00
This statement about matter and its behavior is best classified as a
Answers: 1
You know the right answer?
the molar concentration (m) of a solution prepared by dissolving 0.2362g of cr(no3)3 in a 50-ml volu...
Questions


History, 05.10.2021 06:20

Biology, 05.10.2021 06:20

History, 05.10.2021 06:20




Mathematics, 05.10.2021 06:20



Mathematics, 05.10.2021 06:20

Chemistry, 05.10.2021 06:20

Chemistry, 05.10.2021 06:20


Chemistry, 05.10.2021 06:20

Mathematics, 05.10.2021 06:20

Mathematics, 05.10.2021 06:20



Mathematics, 05.10.2021 06:20