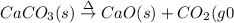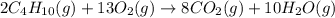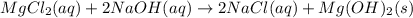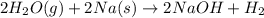Write a balanced molecular equation describing each of the following chemical reactions:
(a)s...

Chemistry, 02.09.2019 20:10 codyshs160
Write a balanced molecular equation describing each of the following chemical reactions:
(a)solid calcium carbonate is heated and decomposes to sold calcium oxide and carbon dioxide
(b)gaseous butane, c4h10, reacts with diatomic oxygen gas to yield gaseous carbon dioxide and water vapor
(c)aaqeous solutions of magnesium chloride and sodium hydroxide react to produce solid magnesium hydroxide and aqueous sodium chloride
(d)water vapor reacts with sodium metal to produce solid sodium hydroxide and hydrogen gas

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:00
Why are the trends and exceptions to the trends in ionization energy observed?
Answers: 1

Chemistry, 22.06.2019 05:30
Transportation is the largest single source of air pollution in the united states. air pollution can harm the environment and human health. which technology could offer a solution to this problem? mufflers that reduce noise motors that run on electricity tires that improve gas mileage
Answers: 3


Chemistry, 23.06.2019 09:00
How many grams of ammonia are produced when 1.0 mole of nitrogen reacts
Answers: 2
You know the right answer?
Questions


Mathematics, 28.08.2019 08:10

Chemistry, 28.08.2019 08:10

English, 28.08.2019 08:10

Mathematics, 28.08.2019 08:10


Social Studies, 28.08.2019 08:10

Mathematics, 28.08.2019 08:10


English, 28.08.2019 08:10

English, 28.08.2019 08:10

Arts, 28.08.2019 08:10

Mathematics, 28.08.2019 08:10

History, 28.08.2019 08:10

English, 28.08.2019 08:10

Computers and Technology, 28.08.2019 08:10


Mathematics, 28.08.2019 08:10


Mathematics, 28.08.2019 08:10