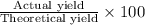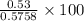Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:20
Calcium hydride (cah2) reacts with water to form hydrogen gas: cah2(s) + 2h2o(l) → ca(oh)2(aq) + 2h2(g) how many grams of cah2 are needed to generate 45.0 l of h2 gas at a pressure of 0.995 atm and a temperature of 32 °c?
Answers: 2

Chemistry, 22.06.2019 00:00
Substance x has a fixed volume, and the attraction between its particles is strong .substance y had widely spread out particles and can be compressed what can most likely be concluded about these substances
Answers: 2

Chemistry, 22.06.2019 00:40
During which time interval does the object travel approximately 10 meters
Answers: 3

Chemistry, 22.06.2019 03:30
Asample of ammonia reacts with oxygen as shown. 4nh3(g) + 5o2(g) 4no(g) + 6h2o(g) what is the limiting reactant if 4.0 g of nh3 react with 8.0 g of oxygen? o2 because it produces only 0.20 mol of no. nh3 because it produces only 0.20 mol of no. o2 because it produces two times less no than nh3. nh3 because it produces three times more no than o2.
Answers: 3
You know the right answer?
Asample of 53.0 of carbon dioxide was obtained by heating 1.31 g of calcium carbont. what is the per...
Questions



English, 19.02.2021 19:30

Mathematics, 19.02.2021 19:30

Social Studies, 19.02.2021 19:30

Health, 19.02.2021 19:30

Mathematics, 19.02.2021 19:30




Mathematics, 19.02.2021 19:30

Mathematics, 19.02.2021 19:30


Mathematics, 19.02.2021 19:30

Mathematics, 19.02.2021 19:30

Mathematics, 19.02.2021 19:30

Physics, 19.02.2021 19:30

English, 19.02.2021 19:30

Computers and Technology, 19.02.2021 19:30