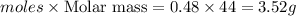Chemistry, 05.09.2019 22:20 coollid876
For the reaction c6h6 + ? o2 → ? co2+? h2o , what is the maximum amount of co2 which could be formed from 6.59 g of c6h6 and 6.62 mol of o2? answer in units of g.

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 04:50
The name of the ion, s2-, is: sulfurous ion sulfide ion sulfur ion sulfate ion
Answers: 1

Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3

Chemistry, 22.06.2019 09:30
Mike and mitchell decide to have a foot race. they mark off a stretch of 100 yards, and recruit cindy to work the stopwatch. after running the race and looking at the results, cindy declared that mitchell was the fastest. so how did the boys times compare?
Answers: 3
You know the right answer?
For the reaction c6h6 + ? o2 → ? co2+? h2o , what is the maximum amount of co2 which could be for...
Questions


History, 25.11.2019 13:31

Health, 25.11.2019 13:31



Geography, 25.11.2019 13:31

Mathematics, 25.11.2019 13:31

Mathematics, 25.11.2019 13:31


Mathematics, 25.11.2019 13:31



Mathematics, 25.11.2019 13:31

Chemistry, 25.11.2019 13:31

Mathematics, 25.11.2019 13:31



History, 25.11.2019 13:31

Geography, 25.11.2019 13:31

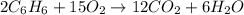
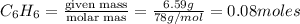
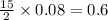 moles of oxygen
moles of oxygen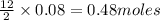 of carbon dioxide
of carbon dioxide