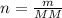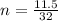Chemistry, 06.09.2019 05:30 rubiabori6579
Achemist measures the amount of oxygen gas produced during an experiment. he finds that 11.5 g of oxygen gas is produced. calculate the number of moles of oxygen gas produced.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:40
In an effort to address concerns about global warming, a power plant in portland,oregon is designed to take all of its exhaust gases from its boilers and recycle the co2 using the solvay process to make sodium hydrogen carbonate. the reaction is shown below. nh3(g) + h2o(l) + co2(g) + nacl(aq) → nahco3(aq) + nh4cl(aq) how many liters each of nh3 and co2 (both at stp) would be consumed to produce 3.00 kg of sodium bicarbonate? the volume of both nh3 and co2 would be
Answers: 1



Chemistry, 22.06.2019 19:30
Anurse used a 0.02-mg/l solution of disinfection to clean a patients wound. what is the concentration of the solution expressed as a percentage?
Answers: 1
You know the right answer?
Achemist measures the amount of oxygen gas produced during an experiment. he finds that 11.5 g of ox...
Questions

History, 31.10.2019 07:31

Social Studies, 31.10.2019 07:31


Chemistry, 31.10.2019 07:31


Chemistry, 31.10.2019 07:31


History, 31.10.2019 07:31

Health, 31.10.2019 07:31



Mathematics, 31.10.2019 07:31

Mathematics, 31.10.2019 07:31


Mathematics, 31.10.2019 07:31





English, 31.10.2019 07:31