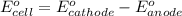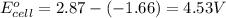Chemistry, 06.09.2019 16:20 glizbethh00
For the following electrochemical reaction: al3+(aq) + 3e -> al(s) eº = -1.66 v e° = 2.87 f2(g) + 2e -> 2f (aq) calculate eº for the cell.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Ibeg i need 20. a reaction produces 4.93 l of oxygen, but was supposed to produce 1 mol of oxygen. what is the percent yield?
Answers: 1

Chemistry, 22.06.2019 03:00
Which best describes how johannes kepler developed his laws of planetary motion
Answers: 3

Chemistry, 22.06.2019 09:00
Chemical energy is a form of a. kinetic energy only. b. both potential and kinetic energy. c. neither potential nor kinetic energy. d. potential energy only. reset
Answers: 1

Chemistry, 22.06.2019 10:50
A100 kmol/h stream that is 97 mole% carbon tetrachloride (ccl4) and 3% carbon disulfide (cs2) is to be recovered from the bottom of a distillation column. the feed to the column is 16 mole% cs2 and 84% ccl4, and 2% of the ccl4 entering the column is contained in the overhead stream leaving the top of the column. calculate the mass and mole fractions of ccl4 in the overhead stream, and determine the molar flow rates of ccl4 and cs2 in the overhead and feed streams. 12. mw_ccla- 153.82; mw_cs2-76.14.
Answers: 3
You know the right answer?
For the following electrochemical reaction: al3+(aq) + 3e -> al(s) eº = -1.66 v e° = 2.87 f2(g)...
Questions

Business, 23.07.2019 16:20

History, 23.07.2019 16:20

Biology, 23.07.2019 16:20


Biology, 23.07.2019 16:20

Biology, 23.07.2019 16:20

Business, 23.07.2019 16:20

History, 23.07.2019 16:20

History, 23.07.2019 16:20

Biology, 23.07.2019 16:20

Biology, 23.07.2019 16:20


History, 23.07.2019 16:30

History, 23.07.2019 16:30

History, 23.07.2019 16:30

History, 23.07.2019 16:30

Mathematics, 23.07.2019 16:30

Biology, 23.07.2019 16:30


Biology, 23.07.2019 16:30

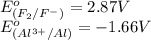
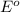 potential will always get reduced and will undergo reduction reaction. Here, fluorine will undergo reduction reaction will get reduced.
potential will always get reduced and will undergo reduction reaction. Here, fluorine will undergo reduction reaction will get reduced.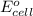 of the reaction, we use the equation:
of the reaction, we use the equation: