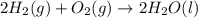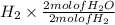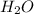Chemistry, 06.09.2019 23:30 quissowavyquis8484
Consider the balanced chemical equation that follows. you are asked to determine how many moles of water you can produce from 4.0 mol of hydrogen and excess oxygen. (excess oxygen means that so much oxygen is available it will not run out.) which of the numbers that appear in the balanced chemical equation below are used to perform this calculation?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:30
1. calculate the approximate enthalpy of the reaction in joules. estimate that 1.0 ml of vinegar has the same thermal mass as 1.0 ml of water. iqnore the thermal mass of th sodium bicarbonate. note: it takes about 4.2 joules () to change 1.0 gram (1.0ml) of water 1.0 c
Answers: 2

Chemistry, 22.06.2019 14:00
Will mark brainliest how many electrons can be held in the energy level n = 4?
Answers: 1

Chemistry, 22.06.2019 23:00
In which region is the substance in both the solid phase and the liquid phase? 1 2. 3 4 mark this and return save and exit next
Answers: 2

Chemistry, 23.06.2019 00:50
What is the enthalpy of combustion (per mole) of c4h10 (g)? –2,657.5 kj/mol –5315.0 kj/mol –509.7 kj/mol –254.8 kj/mol
Answers: 1
You know the right answer?
Consider the balanced chemical equation that follows. you are asked to determine how many moles of w...
Questions

Computers and Technology, 20.07.2019 03:30


Computers and Technology, 20.07.2019 03:30

Mathematics, 20.07.2019 03:30


History, 20.07.2019 03:30

Mathematics, 20.07.2019 03:30



Biology, 20.07.2019 03:30




Social Studies, 20.07.2019 03:30



Mathematics, 20.07.2019 03:30

Mathematics, 20.07.2019 03:30

English, 20.07.2019 03:30