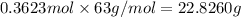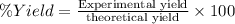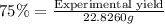Chemistry, 09.09.2019 17:20 victoria1831
If the percent yield for the following reaction is 75.0%, and 25.0 g of no₂ are consumed in the reaction, how many grams of nitric acid, hno₃(aq) are produced?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:30
You are to give ampicillin with a recommended dose of 25mg/kg to a child with a mass of 29kg. if stock on hand is 250mg/capsule how many capsules should be given?
Answers: 1

Chemistry, 22.06.2019 05:00
Cucl2 + 2nano3 cu(no3)2 + 2nacl what is the percent yield of nacl if 31.0 g of cucl2 reacts with excess nano3 to produce 21.2 g of nacl? 49.7% 58.4% 63.6% 78.7%
Answers: 1

Chemistry, 22.06.2019 19:50
A2.5% (by mass) solution concentration signifies that there is a 2.5 % (by mass) solution concentration signifies that there is blank of solute in every 100 g of solution. of solute in every 100 g of solution
Answers: 3

Chemistry, 22.06.2019 22:30
Amedication is given at a dosage of 3.000 mg of medication per kg of body weight. if 0.1500 g of medication is given, then what was the patient's weight in pounds (lbs)? there are 453.59g in 1 lb.
Answers: 2
You know the right answer?
If the percent yield for the following reaction is 75.0%, and 25.0 g of no₂ are consumed in the reac...
Questions


Social Studies, 10.10.2019 22:50

History, 10.10.2019 22:50

English, 10.10.2019 22:50

French, 10.10.2019 22:50

Biology, 10.10.2019 22:50



History, 10.10.2019 22:50

English, 10.10.2019 22:50



Geography, 10.10.2019 22:50




Social Studies, 10.10.2019 22:50



English, 10.10.2019 22:50

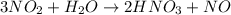
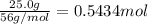
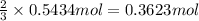 of nitric acid.
of nitric acid.