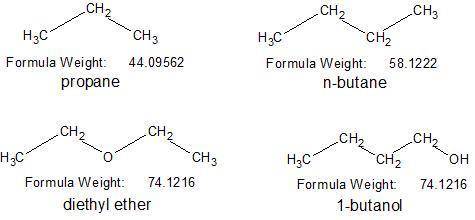
Based on the type or types of intermolecular forces, predict the substance in each pair that has the higher boiling point. match the words in the left column to the appropriate blanks in the sentences on the right. make certain each sentence is complete before submitting your answer. given the molecules propane (c3h8) and n-butane (c4h10), given the molecules propane ( c 3 h 8 ) and n-butane ( c 4 h 10 ) , blank has the higher boiling point mainly due to blank. has the higher boiling point mainly due to given the molecules propane ( c 3 h 8 ) and n-butane ( c 4 h 10 ) , blank has the higher boiling point mainly due to blank.. given the molecules diethyl ether (ch3ch2och2ch3) and 1-butanol (ch3ch2ch2ch2oh), given the molecules diethyl ether ( c h 3 c h 2 o c h 2 c h 3 ) and 1-butanol ( c h 3 c h 2 c h 2 c h 2 o h ) , blank has the higher boiling point mainly due to blank. has the higher boiling point mainly due to given the molecules diethyl ether ( c h 3 c h 2 o c h 2 c h 3 ) and 1-butanol ( c h 3 c h 2 c h 2 c h 2 o h ) , blank has the higher boiling point mainly due to blank..

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:40
The formation of a solid, also known as a is an indication of a chemical change. precipitate particulate particle powder
Answers: 3

Chemistry, 22.06.2019 20:30
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 3

Chemistry, 23.06.2019 00:00
How is the way a mixture is combined different from how a compound is combined?
Answers: 3

Chemistry, 23.06.2019 02:20
Why dose heating increase the speed at which a solution dissolved in water
Answers: 1
You know the right answer?
Based on the type or types of intermolecular forces, predict the substance in each pair that has the...
Questions


Physics, 24.04.2020 03:18

English, 24.04.2020 03:18





History, 24.04.2020 03:20


Mathematics, 24.04.2020 03:20

English, 24.04.2020 03:20

Mathematics, 24.04.2020 03:20


Mathematics, 24.04.2020 03:20

Mathematics, 24.04.2020 03:20

Advanced Placement (AP), 24.04.2020 03:20