
Rate law equation the rate of a chemical reaction depends on the concentrations of the reactants. for the general reaction between a and b, aa+bb⇌cc+dd the dependence of the reaction rate on the concentration of each reactant is given by the equation called the rate law: rate=k[a]m[b]n where k is a proportionality constant called the rate constant. the exponent m determines the reaction order with respect to a, and n determines the reaction order with respect to b. the overall reaction order equals the sum of the exponents (m+n). part a part complete what is the reaction order with respect to a? express your answer as an integer.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 12:00
What is the lowest number energy level where a d sublevel is found
Answers: 1

Chemistry, 22.06.2019 12:30
When a scientific theory has been tested and proved by the scientific community, it becomes a law
Answers: 2


Chemistry, 23.06.2019 11:30
Place the following substances in order of ph from lowest ph to highest. a. neutral compounds, bases, acids b. acids, neutral compounds, bases c. bases, acids, neutral compounds d. bases, neutral compounds, acids
Answers: 1
You know the right answer?
Rate law equation the rate of a chemical reaction depends on the concentrations of the reactants. fo...
Questions



Social Studies, 22.07.2019 04:10




History, 22.07.2019 04:10







Business, 22.07.2019 04:10



History, 22.07.2019 04:10

Chemistry, 22.07.2019 04:10

Biology, 22.07.2019 04:10

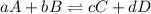
![Rate=k[A]^m[B]^n](/tpl/images/0226/8493/b2185.png)



