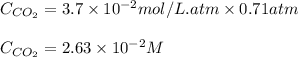The partial pressure of co2 gas above the liquid in a carbonated drink is 0.71 atm. assuming that the henry's law constant for co2 in the drink is that same as that in water, 3.7 x 10-2 mol/l atm, calculate the solubility of carbon dioxide in this drink. give your answer to 3 decimal places.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 12:00
An atom of which element reacts with an atom of hydrogen to form a bond with the greatest degree of polarity ?
Answers: 1

Chemistry, 22.06.2019 13:50
How does the motion of particles in a gas change as the gas cools
Answers: 2

Chemistry, 22.06.2019 18:00
How does climate change cause the ocean's thermohaline current to slow down?
Answers: 3

Chemistry, 22.06.2019 21:00
How many neutrons does an element have if its atomic number is 50 and its mass number is 166
Answers: 1
You know the right answer?
The partial pressure of co2 gas above the liquid in a carbonated drink is 0.71 atm. assuming that th...
Questions

Advanced Placement (AP), 14.02.2021 05:40

Mathematics, 14.02.2021 05:40

Mathematics, 14.02.2021 05:40

History, 14.02.2021 05:40


Mathematics, 14.02.2021 05:40


Health, 14.02.2021 05:40



Arts, 14.02.2021 05:40


History, 14.02.2021 05:40





Mathematics, 14.02.2021 05:40

Social Studies, 14.02.2021 05:40

Spanish, 14.02.2021 05:40

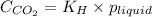
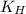 = Henry's constant =
= Henry's constant = 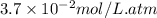
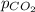 = partial pressure of carbonated drink = 0.71 atm
= partial pressure of carbonated drink = 0.71 atm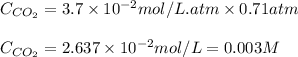
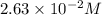
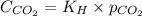
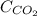 = molar solubility of carbon dioxide gas = ?
= molar solubility of carbon dioxide gas = ?