

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 21:30
Liquid ammonia is produced at high temperatures and under great pressure in a tank by passing a mixture of nitrogen gas and hydrogen gas over an iron catalyst. the reaction is represented by the following equation. n2(g) + 3h2(g) → 2nh3(g) changing all but one experimental condition will affect the amount of ammonia produced. that condition is a) increasing the concentration of both reactants b) changing the temperature within the tank c) decreasing the pressure within the tank. d) increasing only the amount of nitrogen present.
Answers: 1

Chemistry, 23.06.2019 04:20
The graph shows one consequence of urban sprawl. how did urban sprawl contribute to the change in biodiversity
Answers: 2

Chemistry, 23.06.2019 17:00
Liquid nitrogen is kept at a temperature of -320 degrees. when liquid nitrogen is heated it quickly boils and turns into gas. which pair of pictures represent the change caused by adding heat to liquid nitrogen?
Answers: 3
You know the right answer?
You are given 12.33 moles of o2. how many moles of co2 can be made?...
Questions

Business, 22.11.2020 06:40

Mathematics, 22.11.2020 06:40


Mathematics, 22.11.2020 06:40

Mathematics, 22.11.2020 06:40

History, 22.11.2020 06:40

Chemistry, 22.11.2020 06:40

Social Studies, 22.11.2020 06:40

Mathematics, 22.11.2020 06:40

Biology, 22.11.2020 06:40

Mathematics, 22.11.2020 06:40

English, 22.11.2020 06:40

Physics, 22.11.2020 06:40

Mathematics, 22.11.2020 06:40




Social Studies, 22.11.2020 06:40

Mathematics, 22.11.2020 06:40

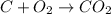
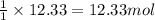 of carbon dioxide gas.
of carbon dioxide gas.

