
Chemistry, 14.09.2019 09:10 robert7248
Asample of an oxide of antimony (sb) contain 19.75 g of antimony combine with 6.5 g of oxygen . what is the simplest formula for the oxide

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 11:00
3) in peaches, [oh]=3.16x10-11 m a) find [h+ ] b) what is the ph? c) is the solution acidic, basic, or neutral?
Answers: 1


Chemistry, 22.06.2019 12:10
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 14:50
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of tthe table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3
You know the right answer?
Asample of an oxide of antimony (sb) contain 19.75 g of antimony combine with 6.5 g of oxygen . what...
Questions


Computers and Technology, 10.02.2021 19:30

Mathematics, 10.02.2021 19:30

English, 10.02.2021 19:30




English, 10.02.2021 19:30

History, 10.02.2021 19:30

Mathematics, 10.02.2021 19:30

Mathematics, 10.02.2021 19:30


Mathematics, 10.02.2021 19:30

Mathematics, 10.02.2021 19:30


Mathematics, 10.02.2021 19:30



Mathematics, 10.02.2021 19:30

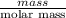
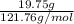
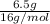
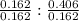
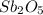 .
.

