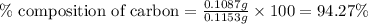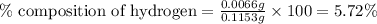Chemistry, 14.09.2019 09:30 bagofmud8339
A0.1153-gram sample of a pure hydrocarbon was burned in a c-h combustion train to produce 0.3986 gram of co2and 0.0578 gram of h2o. determine the masses of c and h in the sample and the percentages of these elements in this hydrocarbon.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:00
Write a hypothesis that answers the lesson question, “while observing a chemical reaction, how can you tell which reactant is limiting? ” hypothesis: if a substance is the limiting reactant, then . . because . .
Answers: 1

Chemistry, 22.06.2019 08:00
This classification of drug typically changes the brain's chemistry and reduces its ability to create its own endorphins.
Answers: 1

Chemistry, 22.06.2019 11:00
Ais a mountain created from eruptions of lava, ash, rocks, and hot gases.
Answers: 1

Chemistry, 22.06.2019 18:00
Answer asap need it by wednesday morning carry out the following calculations on ph and ka of from data. i. calculate the ph of 0.02m hcl ii. calculate the ph of 0.036m naoh iii. calculate the ph of 0.36m ca(oh)2 iv. calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 v. calculate ka for weak acid ha which has a ph of 3.65 at 0.30m concentration vi. calculate the ka of a solution made by mixing 15.0 cm3 0.2m ha and 60.0 cm3 0.31m a-. [ph= 3.80] vii. calculate the ph of a solution made by mixing 15.0 cm3 0.1m naoh and 35.0 cm3 0.2m hcooh. [ka = 1.82 x 10-4 m]
Answers: 1
You know the right answer?
A0.1153-gram sample of a pure hydrocarbon was burned in a c-h combustion train to produce 0.3986 gra...
Questions



English, 29.07.2019 06:30


English, 29.07.2019 06:30



History, 29.07.2019 06:30



History, 29.07.2019 06:30

English, 29.07.2019 06:30



English, 29.07.2019 06:30

Social Studies, 29.07.2019 06:30

English, 29.07.2019 06:30



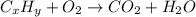
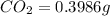
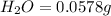
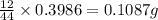 of carbon will be contained.
of carbon will be contained.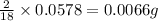 of hydrogen will be contained.
of hydrogen will be contained.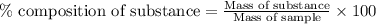 ......(1)
......(1)