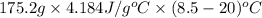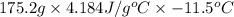Chemistry, 18.09.2019 04:10 lmoleary7466
Asample of ammonium nitrate, weighing 25.2 g, was added to a 150.0 ml container of h2o at 20.0 °c, and then is thoroughly dissolved. the final temperature of the mixture is 8.5 °c. find how much heat (kj) was lost/gained by the solution (surroundings).

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:10
How do forces between particles in gases compare to forces in the other states of matter? o a. the forces in gases are stronger than forces in solids but weaker than forces in liquids. o b. the forces in gases are weaker than forces in solids but stronger than forces in liquids. o c. the forces in gases are weaker than forces in solids and liquids. o d. the forces in gases are stronger than forces in solids and liquids. submit
Answers: 1

Chemistry, 22.06.2019 03:30
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 2

Chemistry, 22.06.2019 07:00
Indicate whether the specified alkyl halides will form primarily substitution products, only elimination products, both substitution and elimination products, or no products when they react with sodium methoxide. 1-bromobutane 1-bromo-2-methylpropane 2-bromobutane 2-bromo-2-methylpropane
Answers: 2

Chemistry, 22.06.2019 10:30
Use this information to determine the number of calends electrons in the atoms. which of the following correctly compares the stability of the two atoms? a) both are unreactive b) both are highly reactive c) a is unreactive and d is reactive d) a is reactive and d is unreactive
Answers: 2
You know the right answer?
Asample of ammonium nitrate, weighing 25.2 g, was added to a 150.0 ml container of h2o at 20.0 °c, a...
Questions


Mathematics, 07.10.2020 08:01

Mathematics, 07.10.2020 08:01

English, 07.10.2020 08:01

Mathematics, 07.10.2020 08:01

Social Studies, 07.10.2020 08:01

Mathematics, 07.10.2020 08:01

English, 07.10.2020 08:01



Social Studies, 07.10.2020 08:01



English, 07.10.2020 08:01

Mathematics, 07.10.2020 08:01


Mathematics, 07.10.2020 08:01

Chemistry, 07.10.2020 08:01

Mathematics, 07.10.2020 08:01

English, 07.10.2020 08:01

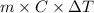
 = change in temperature
= change in temperature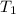 is
is 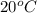 ,
, 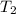 is
is 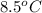 , and specific heat of water is 4.184
, and specific heat of water is 4.184 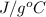 .
.