Chemistry, 18.09.2019 05:30 asuhdude57
A245.7g sample of metal at 75.0℃ was placed in 115.4g of water at 22.0℃. the final temperature of the water and metal was 34.0℃. if no heat was lost to the surroundings, what is the specific heat of the metal? (specific heat of water = 4.184 j/g℃)

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Write the symbol for every chemical element that has atomic number greater than 3 and atomic mass less than 12.0 u.
Answers: 1


Chemistry, 22.06.2019 08:30
What method(s) do plants use to obtain nitrogen? select all that apply. absorb it from the atmosphere use bacteria to convert nitrogen to usable form obtain usable nitrogen compounds from the soil absorb nitrogen from water taken in at the roots
Answers: 3

You know the right answer?
A245.7g sample of metal at 75.0℃ was placed in 115.4g of water at 22.0℃. the final temperature of th...
Questions

Chemistry, 04.02.2021 04:50




Mathematics, 04.02.2021 04:50

Mathematics, 04.02.2021 04:50

Mathematics, 04.02.2021 04:50




Advanced Placement (AP), 04.02.2021 04:50

Mathematics, 04.02.2021 04:50

Mathematics, 04.02.2021 04:50

English, 04.02.2021 04:50




Mathematics, 04.02.2021 04:50

English, 04.02.2021 04:50

Mathematics, 04.02.2021 04:50

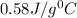
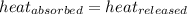
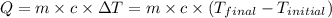
![m_1\times c_1\times (T_{final}-T_1)=-[m_2\times c_2\times (T_{final}-T_2)]](/tpl/images/0238/8184/09236.png) .................(1)
.................(1)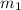 = mass of metal = 245.7 g
= mass of metal = 245.7 g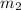 = mass of water = 115.4 g
= mass of water = 115.4 g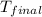 = final temperature =
= final temperature = 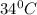
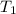 = temperature of metal =
= temperature of metal = 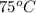
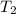 = temperature of water =
= temperature of water = 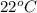
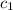 = specific heat of metal = ?
= specific heat of metal = ?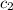 = specific heat of water=
= specific heat of water= 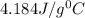
![-245.7\times c\times (75-34)=[115.4\times 4.184\times (34-22)]](/tpl/images/0238/8184/37846.png)