
Polonium- 210 , po210 , decays to lead- 206 , pb206 , by alpha emission according to the equation po84210⟶pb82206+he24 if the half-life, /2 , of po210 is 138.4 days , calculate the mass of pb206 produced from a 561.0 mg sample of polonium(iv) chloride, pocl4 , that is left untouched for 333.8 days . assume that the only polonium isotope present in the sample is the po210 isotope. the isotopic molar masses of po210 is 209.98 g/mol and pb206 is 205.97 g/mol .

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Which type of bond is present in hydrogen sulfide (h2s)? the table of electronegativities is given. a. hydrogen b. ionic c. nonpolar covalent d. polar covalent
Answers: 1

Chemistry, 22.06.2019 09:00
Ineed to find the answer of this question because i dont understand it
Answers: 1

Chemistry, 22.06.2019 14:50
Which of the following is most likely true about water in chemical solutions?
Answers: 1

Chemistry, 22.06.2019 15:00
Describe what happens to the molecules as water goes from ice to liquid to vapor. be sure to explain what happens to the temperature during the phase changes.
Answers: 2
You know the right answer?
Polonium- 210 , po210 , decays to lead- 206 , pb206 , by alpha emission according to the equation po...
Questions

Biology, 07.01.2020 18:31

Mathematics, 07.01.2020 18:31


Biology, 07.01.2020 18:31

Mathematics, 07.01.2020 18:31



English, 07.01.2020 18:31


English, 07.01.2020 18:31

English, 07.01.2020 18:31



Mathematics, 07.01.2020 18:31


Biology, 07.01.2020 18:31

Business, 07.01.2020 18:31


Mathematics, 07.01.2020 18:31

Mathematics, 07.01.2020 18:31

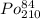 ⟶
⟶ 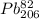 +
+ 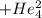
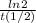
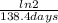 =0,005008 days^(-1)
=0,005008 days^(-1)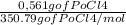 = 0,001599 mol of PoCl4
= 0,001599 mol of PoCl4

