Chemistry, 25.09.2019 01:00 vinp190p9zekn
One mole of pure n2 and 1 mole of pure o2 are contained in separate compartments of a rigid and insulated container at 1 bar and 298 k. the separator between the compartments are later removed to allow the mixing of the gases. assume that both gases are in the ideal gas state.
a) what is the final t and p of the mixture?
b) for a mixture of ideal gases, each component carries a partial pressure that is proportional to its mole fraction: i. e., = p and = p, p being the total pressure of the mixture. calculate the entropy change of the mixing process and draw a schematic showing the hypothetical path you used for the calculation.
i am unclear why you need enthalpy information when the question asks for entropy. can someone solve part a?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 09:00
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 2

Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3

Chemistry, 23.06.2019 06:10
How can liquids be seperated by density a the liquids are absorbed onto a paper b the liquids are turned into seperate vapors c the liquids are collected as they evaporate d the liquids are allowed to seperate into layers
Answers: 1

Chemistry, 23.06.2019 06:30
Moving force of air flows from areas of high pressure to areas of low pressure true or false
Answers: 2
You know the right answer?
One mole of pure n2 and 1 mole of pure o2 are contained in separate compartments of a rigid and insu...
Questions


Mathematics, 04.10.2020 18:01


Mathematics, 04.10.2020 18:01

English, 04.10.2020 18:01


Social Studies, 04.10.2020 18:01

Mathematics, 04.10.2020 18:01

Mathematics, 04.10.2020 18:01



History, 04.10.2020 18:01

Mathematics, 04.10.2020 18:01




Computers and Technology, 04.10.2020 19:01

Mathematics, 04.10.2020 19:01


Advanced Placement (AP), 04.10.2020 19:01

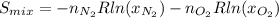
![S_{mix}=-(1mol)[(8.314J/(mol*K)]ln(0.5)-(1mol)[(8.314J/(mol*K)]ln(0.5)](/tpl/images/0259/8106/ed779.png)