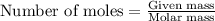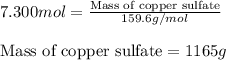Chemistry, 25.09.2019 02:00 bluenblonderw
Achemical process requires 7.300 g-mol of calcium sulfate (caso.) for a reaction. how many grams are in 7.300 g-mol of calcium sulfate? caso

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 04:40
*will mark you brainliest + 15 points ** why does the equilibrium of a system shift when the pressure is increased? a. to maximize the stress on the system b. to stop restoring equilibrium to the system c. to increase the total moles of gas in the system d. to decrease the total moles of gas in the system
Answers: 3

Chemistry, 22.06.2019 17:10
Acalorimeter is to be calibrated: 51.203 g of water at 55.2 degree c is added to a calorimeter containing 49.783 g of water at 23.5c. after stirring and waiting for the system to equilibrate, the final temperature reached is 37.6 degree c. specific heat capacity of water (s = 4.18 j/g∙degree c). calculate the calorimeter constant. (smδt)warm water = -[(smδt)cold water + (calorimeterδtcold water)]
Answers: 2

Chemistry, 23.06.2019 00:00
What is the pressure of 0.500 moles of carbon dioxide gas in a 2.5 l tank and at a temperature of 301 k? (r=0.0821 l·atm/mol·k) 3.08 atm 1.2 atm 0.23 atm 4.01 atm 4.94 atm
Answers: 1
You know the right answer?
Achemical process requires 7.300 g-mol of calcium sulfate (caso.) for a reaction. how many grams are...
Questions

Mathematics, 04.09.2020 07:01

Mathematics, 04.09.2020 07:01

Computers and Technology, 04.09.2020 07:01

Mathematics, 04.09.2020 07:01

English, 04.09.2020 07:01

Mathematics, 04.09.2020 07:01



Geography, 04.09.2020 07:01




English, 04.09.2020 07:01


Mathematics, 04.09.2020 07:01


Mathematics, 04.09.2020 07:01

Social Studies, 04.09.2020 07:01

Mathematics, 04.09.2020 07:01

Arts, 04.09.2020 07:01