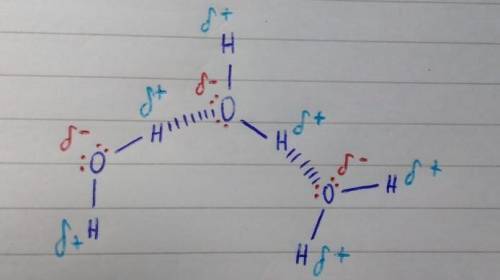
Chemistry, 02.10.2019 20:30 sativataurus
Raw three water molecules interacting with each other. indicate hydrogen-bonding with dashed lines. show lone pairs!

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 15:00
Which element in the third period would you expect to have the larger atomic radius, sodium (na) or sulfur (s)? a. sodium, because it has a higher effective nuclear charge attracting electrons in fewer energy levels. b. sodium, because it has fewer protons attracting electrons in the same energy levels. c. sulfur, because it has more protons attracting electrons in more energy levels. d. sulfur, because it has a higher effective nuclear charge attracting electrons in the same energy levels.
Answers: 2

Chemistry, 23.06.2019 02:00
Which of the following substances is the most soluble in water? a. sodium chloride b. methane c. bromine d. carbon
Answers: 1

Chemistry, 23.06.2019 05:00
Select the statement that describe chemical properties a. antacid tablets neutralize stomach acid b. helium is the lightest monatomic element c. water freezes at 0 celsius d. mercury is liquid at room temperature
Answers: 3

Chemistry, 23.06.2019 09:20
Four statements about the development of the atomic model are shown below. a: electrons have wavelike properties. b: atoms have small, negatively charged particles. c. the center of an atom is a small, dense nucleus. d: atoms are hard, indivisible spheres. which order of statements represents the historical development of the atomic model? c-d-a-b c-d-b-a d— в-а — с d-b-c-a
Answers: 1
You know the right answer?
Raw three water molecules interacting with each other. indicate hydrogen-bonding with dashed lines....
Questions




Chemistry, 24.09.2020 21:01

Mathematics, 24.09.2020 21:01

Mathematics, 24.09.2020 21:01

History, 24.09.2020 21:01

Mathematics, 24.09.2020 21:01

History, 24.09.2020 21:01

Mathematics, 24.09.2020 21:01

Physics, 24.09.2020 21:01






Mathematics, 24.09.2020 21:01



English, 24.09.2020 21:01