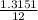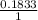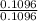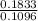Chemistry, 06.10.2019 00:00 Savadt2810
When a sample of cyclohexene is burned in oxygen, 4.822 g of co2 and 1.650 g of h2o are obtained. what is the simplest formula for cyclohexene?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 10:00
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1


Chemistry, 22.06.2019 17:10
)benzene and toluene form nearly ideal solutions. consider an equimolar solution of benzene and toluene. at 20 °c the vapour pressures of pure benzene and toluene are 9.9 kpa and 2.9 kpa, respectively. the solution is boiled by reducing the external pressure below the vapour pressure. calculate (i) the pressure when boiling begins, (ii) the composition of each component in the vapour, and (iii) the vapour pressure when only a few drops of liquid remain. assume that the rate of vaporization is low enough for the temperature to remain constant at 20 °c.
Answers: 1

Chemistry, 22.06.2019 19:30
Helium decays to form lithium. which equation correctly describes this decay?
Answers: 2
You know the right answer?
When a sample of cyclohexene is burned in oxygen, 4.822 g of co2 and 1.650 g of h2o are obtained. wh...
Questions


Mathematics, 24.11.2020 21:40






History, 24.11.2020 21:40

English, 24.11.2020 21:40

Mathematics, 24.11.2020 21:40


Social Studies, 24.11.2020 21:40



Mathematics, 24.11.2020 21:40


Mathematics, 24.11.2020 21:40

Mathematics, 24.11.2020 21:40


World Languages, 24.11.2020 21:40

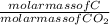 x mass of CO₂
x mass of CO₂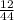 x 4.822 = 1.3151g
x 4.822 = 1.3151g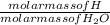 x mass of H₂O
x mass of H₂O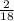 x 1.650 = 0.1833g
x 1.650 = 0.1833g