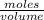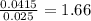Chemistry, 06.10.2019 10:02 Smartpotato9555
Achemist needs to determine the concentration of a sulfuric acid solution by titration with a standard sodium hydroxide solution. he has a 0.1678 m standard sodium hydroxide solution. he takes a 25.00 ml sample of the original acid solution and dilutes it to 250.0 ml. then, he takes a 10.00 ml sample of the dilute acid solution and titrating it with the standard solution. the endpoint was reached after the addition of 19.88 ml of the standard solution. what is the concentration of the original sulfuric acid solution?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:20
Calculate the molarity of 48.0 ml of 6.00 m h2so4 diluted to 0.250 l .
Answers: 1

Chemistry, 22.06.2019 09:00
The nuclear fission process releases neutrons and question 27 options: alpha particles electrons energy beta particles
Answers: 1

Chemistry, 22.06.2019 15:30
The reactions of photosynthesis occur in the of plant cell? a.mitochondria. b. lysosomes. c. chloroplasts. d. chlorophyll
Answers: 1

Chemistry, 22.06.2019 20:00
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1
You know the right answer?
Achemist needs to determine the concentration of a sulfuric acid solution by titration with a standa...
Questions

English, 29.04.2021 16:30


Mathematics, 29.04.2021 16:30

English, 29.04.2021 16:30

Mathematics, 29.04.2021 16:30







Biology, 29.04.2021 16:30


Biology, 29.04.2021 16:30



Mathematics, 29.04.2021 16:30

Chemistry, 29.04.2021 16:30

Chemistry, 29.04.2021 16:30

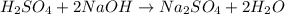
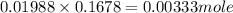
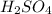 in 10 mL of diluted solution = 1/2 x moles of NaOH
in 10 mL of diluted solution = 1/2 x moles of NaOH
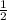 x 0.00333 = 0.00166 mol
x 0.00333 = 0.00166 mol
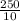 x 0.00166 = 0.0415 mol
x 0.00166 = 0.0415 mol