
Consider a mixture of air and gasoline vapor in a cylinder with a piston. the original volume is 42 cm3. if the combustion of this mixture releases 927 j of energy, to what volume (in l) will the gases expand against a constant pressure of 656 torr if all the energy of combustion is converted into work to push back the piston?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
For the following, determine the type of reaction and then give products.
Answers: 2

Chemistry, 22.06.2019 08:30
Identify one disadvantage to each of the following models of electron configuration: -dot structures -arrow and line diagrams -written electron configurations type in your answer below. (answer) -dot structures do not show the distribution of electrons in orbitals and take up a lot of space. -arrow and line diagrams take up a lot of space and make it difficult to count electrons. -written configurations make it easy to lose count of electrons and do not show the distribution of electrons in orbitals.
Answers: 3

Chemistry, 22.06.2019 14:30
1) describe the physical layout of the ocean floor ? 2) explain how the dumbo octopus swims differently than other octopus species and why this would be an advantage in the aphonic zone . 3) why are the types of organisms that live at each underwater hot vent so dramatically different ?
Answers: 3

Chemistry, 22.06.2019 15:00
Large helium-filled balloons are used to lift scientific equipment to high altitudes. what is the pressure inside such a balloon if it starts out at sea level with a temperature of 10.0ºc and rises to an altitude where its volume is twenty times the original volume and its temperature is – 50.0ºc ?
Answers: 2
You know the right answer?
Consider a mixture of air and gasoline vapor in a cylinder with a piston. the original volume is 42...
Questions

Mathematics, 26.06.2019 02:00




Mathematics, 26.06.2019 02:00


Mathematics, 26.06.2019 02:00




Geography, 26.06.2019 02:00

Biology, 26.06.2019 02:00

Mathematics, 26.06.2019 02:00


Mathematics, 26.06.2019 02:00

Chemistry, 26.06.2019 02:00


Social Studies, 26.06.2019 02:00


Mathematics, 26.06.2019 02:00

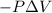
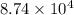 Pa (as 1 torr = 133.3 Pa)
Pa (as 1 torr = 133.3 Pa)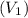 = 42
= 42 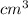
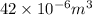 (as 1 m = 100 cm)
(as 1 m = 100 cm)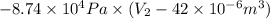
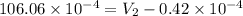
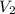 =
= 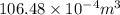
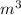 = 1000 L)
= 1000 L)

