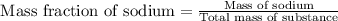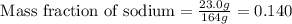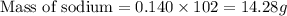Chemistry, 10.10.2019 04:20 villafana36
Asubstance contains 23.0 g sodium, 27.0 g aluminum, and 114 g fluorine. how many grams of sodium are there in a 102-g sample of the substance?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 18:00
How many moles of oxygen gas are produced from the decomposition of six moles of potassium
Answers: 1

Chemistry, 22.06.2019 20:30
Identify the correct mole ratio for each substance. sodium chloride (nacl) na: cl = 1: ammonium nitrate (nhno) h: o = 4:
Answers: 1

Chemistry, 22.06.2019 22:30
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3

Chemistry, 22.06.2019 23:30
To find the work done, the force exerted and distance moved are multiplied. a couch is moved twice before you are happy with its placement. the same force was used to move the couch both times. if more work is done the first time it is moved, what do you know about the distance it was moved? a) when more work was done, the couch was moved the same distance. b) when more work was done, the couch was moved less. c) when more work was done, the couch was moved further. d) when more work was done, the couch wasn't moved at all.
Answers: 1
You know the right answer?
Asubstance contains 23.0 g sodium, 27.0 g aluminum, and 114 g fluorine. how many grams of sodium are...
Questions




Chemistry, 08.04.2020 20:38


Physics, 08.04.2020 20:38



History, 08.04.2020 20:38

Social Studies, 08.04.2020 20:38

Mathematics, 08.04.2020 20:38

Social Studies, 08.04.2020 20:38

Geography, 08.04.2020 20:38




History, 08.04.2020 20:38



Mathematics, 08.04.2020 20:38