
The sulfur content of an ore is determined gravimetrically by reacting the ore with concentrated nitric acid and potassium chlorate, converting all sulfur to sulfate. the excess nitrate and chlorate is removed by reaction with concentrated hydrochloric acid and the sulfate is precipitated using barium cation.
ba2+ (aq) + so42- (aq) = baso4 (s)
analysis of 12.3430 grams of a sulfur containing ore yielded 12.5221 grams of baso4. what is the percent by mass sulfur in the ore? (baso4 = 233.43 g/mol). show all calculation.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 11:00
Which statement correctly identifies the scientific question and describes why the question is scientific? question 1 refers to the supernatural.question 2 reflects a moral or social value.question 3 refers to something that can be measured.question 4 reflects a question that can’t be observed.
Answers: 1

Chemistry, 23.06.2019 01:30
At a certain temperature the rate of this reaction is first order in hi with a rate constant of : 0.0632s2hig=h2g+i2g suppose a vessel contains hi at a concentration of 1.28m . calculate how long it takes for the concentration of hi to decrease to 17.0% of its initial value. you may assume no other reaction is important. round your answer to 2 significant digits.
Answers: 1

Chemistry, 23.06.2019 04:31
Pls i will do pls imma diewhat forms white light? (4 points)a. combination of all wavelengths of ultraviolet light b. combination of all wavelengths of visible lightc. absorption of electromagnetic waves d. absorption of infrared rays
Answers: 2

You know the right answer?
The sulfur content of an ore is determined gravimetrically by reacting the ore with concentrated nit...
Questions

Mathematics, 14.09.2020 16:01

Mathematics, 14.09.2020 16:01

Mathematics, 14.09.2020 16:01

Mathematics, 14.09.2020 16:01

Mathematics, 14.09.2020 16:01

Mathematics, 14.09.2020 16:01

Mathematics, 14.09.2020 16:01

Social Studies, 14.09.2020 16:01

Geography, 14.09.2020 16:01

Social Studies, 14.09.2020 16:01

English, 14.09.2020 16:01

Mathematics, 14.09.2020 16:01

Mathematics, 14.09.2020 16:01

Mathematics, 14.09.2020 16:01

Mathematics, 14.09.2020 16:01

English, 14.09.2020 16:01

Mathematics, 14.09.2020 16:01

English, 14.09.2020 16:01

English, 14.09.2020 16:01

Mathematics, 14.09.2020 16:01

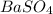 = 12.5221 g
= 12.5221 g
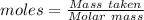
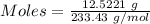
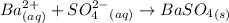
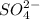
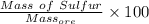 =
= 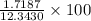 = 13.92 %
= 13.92 %

