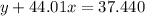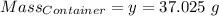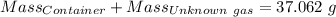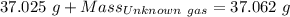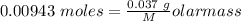Chemistry, 23.10.2019 20:30 camirialchambers17
Agas cylinder filled with nitrogen at standard temperature and pressure has a mass of 37.289 g. the same container filled with carbon dioxide at stp has a mass of 37.440 g. when filled with an unknown gas at stp, the container mass is 37.062 g. calculate the molecular weight of the unknown gas, and then state its probable identity.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 06:30
Identify the missing numbers below to show the result of multiplying the numbers (1.6 × 10-19)(5.0 × 106) = c × 10d
Answers: 1

Chemistry, 22.06.2019 10:10
What shape would a molecule with two bound groups and two lone pairs have?
Answers: 1

Chemistry, 22.06.2019 15:30
Which suspect most likely committed the robbery and how do you know
Answers: 2
You know the right answer?
Agas cylinder filled with nitrogen at standard temperature and pressure has a mass of 37.289 g. the...
Questions

Mathematics, 13.12.2021 17:00

English, 13.12.2021 17:00

Mathematics, 13.12.2021 17:00





SAT, 13.12.2021 17:00

SAT, 13.12.2021 17:00


SAT, 13.12.2021 17:00





Biology, 13.12.2021 17:10


Computers and Technology, 13.12.2021 17:10


Geography, 13.12.2021 17:10

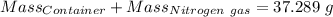
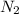 = 28.014 g/mol
= 28.014 g/mol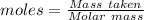
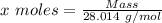
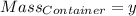
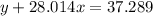
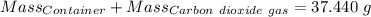
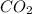 = 44.01 g/mol
= 44.01 g/mol