
Chemistry, 24.10.2019 17:43 odetteerivera
In the process for the manufacture of chlorine by direct oxidation of hcl with air over a catalyst to form cl2 and h2o (only), the exit product is composed of mole fractions of cl2 (21.4%), h2o (21.4%), and n2 (45.0%) – the remainder is hcl and o2. calculate and /or identify: (a) the limiting reactant? (b) the percentage excess reactant? (c) the degree of completion of the reaction? (d) the extent of reaction?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:00
Achef makes salad dressing by mixing oil, vinegar, and spices, as shown. which type of matter is the salad dressing?
Answers: 1

Chemistry, 22.06.2019 10:30
Acompound has a molar mass of 92.02 grams/mole, and its percent composition is 30.4% nitrogen (n) and 69.6% oxygen (o). what is its molecular formula? a. n2o4 b. no2 c. n2o d. n4o2
Answers: 1

Chemistry, 22.06.2019 19:20
Anyone who's in connections academy chemistry b have the factors that affect the rate of a reaction portfolio already done?
Answers: 3

Chemistry, 22.06.2019 20:30
The activation energy for the reaction no2(g)+co2(g)⟶no(g)+co(g) is ea = 300 kj/mol and the change in enthalpy for the reaction is δh = -100 kj/mol . what is the activation energy for the reverse reaction?
Answers: 3
You know the right answer?
In the process for the manufacture of chlorine by direct oxidation of hcl with air over a catalyst t...
Questions


Mathematics, 11.07.2019 08:30

Advanced Placement (AP), 11.07.2019 08:30


Advanced Placement (AP), 11.07.2019 08:30

Mathematics, 11.07.2019 08:30

History, 11.07.2019 08:30

English, 11.07.2019 08:30

Biology, 11.07.2019 08:30

Chemistry, 11.07.2019 08:30


History, 11.07.2019 08:30


Biology, 11.07.2019 08:30

Mathematics, 11.07.2019 08:30

Biology, 11.07.2019 08:30

Mathematics, 11.07.2019 08:30

History, 11.07.2019 08:30


History, 11.07.2019 08:30

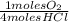 = 0,10725 moles of O₂
= 0,10725 moles of O₂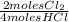 = 0,2145 moles of Cl₂ that are the same than moles of H₂O.
= 0,2145 moles of Cl₂ that are the same than moles of H₂O.
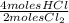 = 0,001 moles of HCl. As initial moles are 0,429 and stoichiometric coefficient is 4, extent of reaction is 0,107
= 0,001 moles of HCl. As initial moles are 0,429 and stoichiometric coefficient is 4, extent of reaction is 0,107

