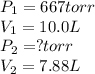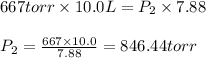Chemistry, 28.10.2019 13:31 floreschachi8230
Fixed amount of gas at 25.0 °c occupies a volume of 10.0 l when the pressure is 667 torr. use boyle's law to calculate the pressure (torr) when the volume is reduced to 7.88 l at a constant temperature of 25.0°c

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 23:30
For the following dehydrohalogenation (e2) reaction, draw the zaitsev product(s) resulting from elimination involving c3–c4 (i.e., the carbon atoms depicted with stereobonds). show the product stereochemistry clearly. if there is more than one organic product, both products may be drawn in the same box. ignore elimination involving c3 or c4 and any carbon atom other than c4 or c3.
Answers: 3

Chemistry, 22.06.2019 00:00
Substance x has a fixed volume, and the attraction between its particles is strong .substance y had widely spread out particles and can be compressed what can most likely be concluded about these substances
Answers: 2

Chemistry, 22.06.2019 14:00
How is the atomic number of a nucleus changed by alpha decay
Answers: 2

Chemistry, 23.06.2019 04:10
Two solids are mixed in a flask and stirred. after a few minutes, the flask becomes cold. which of the following best describes this reaction? a. an exothermic reaction b. a combustion reaction c. an endothermic reaction d. a decomposition reaction
Answers: 1
You know the right answer?
Fixed amount of gas at 25.0 °c occupies a volume of 10.0 l when the pressure is 667 torr. use boyle'...
Questions


English, 23.08.2019 19:30


Physics, 23.08.2019 19:30

Geography, 23.08.2019 19:30

Mathematics, 23.08.2019 19:30

History, 23.08.2019 19:30




Mathematics, 23.08.2019 19:30

Business, 23.08.2019 19:30

English, 23.08.2019 19:30




History, 23.08.2019 19:30


Mathematics, 23.08.2019 19:30

Mathematics, 23.08.2019 19:30

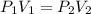
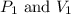 are initial pressure and volume of the gas.
are initial pressure and volume of the gas.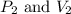 are final pressure and volume of the gas
are final pressure and volume of the gas