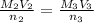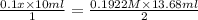Chemistry, 30.10.2019 00:31 tyijiapostell
Achemist needs to determine the concentration of a sulfuric acid solution by titration with a standard sodium hydroxide solution. he has a 0.1922 m standard sodium hydroxide solution. he takes a 25.00 ml sample of the original acid solution and dilutes it to 250.0 ml. then, he takes a 10.00 ml sample of the dilute acid solution and titrates it with the standard solution. the endpoint was reached after the addition of 13.68 ml of the standard solution. what is the concentration of the original sulfuric acid solution?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 07:00
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3

Chemistry, 22.06.2019 10:50
How many grams of oxygen gas are contained in a 15 l sample at 1.02 atm and 28°c? show your work.
Answers: 1

Chemistry, 22.06.2019 14:30
In water, a strong acid will break down into its component parts. a. completely b. partly c. never in water, a weak base will break down into its component parts. a. completely b. partly c. never
Answers: 2

You know the right answer?
Achemist needs to determine the concentration of a sulfuric acid solution by titration with a standa...
Questions

Mathematics, 08.06.2021 05:30


Mathematics, 08.06.2021 05:30



Mathematics, 08.06.2021 05:30


Mathematics, 08.06.2021 05:30

Mathematics, 08.06.2021 05:30

Mathematics, 08.06.2021 05:30




Mathematics, 08.06.2021 05:30


Social Studies, 08.06.2021 05:30

History, 08.06.2021 05:30

Mathematics, 08.06.2021 05:30

English, 08.06.2021 05:30

World Languages, 08.06.2021 05:30

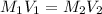
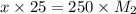
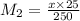
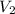 = 10.00 mL (given)
= 10.00 mL (given)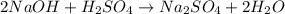
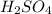 = Concentration × Volume of NaOH
= Concentration × Volume of NaOH