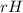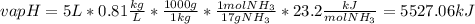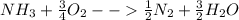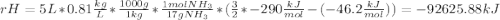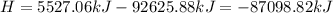Ammonia (nh3) boils at -33∘c; at this temperature it has a density of 0.81 g/cm3. the enthalpy of formation of nh3(g) is -46.2 kj/mol, and the enthalpy of vaporization of nh3(l) is 23.2 kj/mol calculate the enthalpy change when 5 l of liquid nh3 is burned in air to give n2(g) and h2o(g).

Answers: 3


Another question on Chemistry


Chemistry, 21.06.2019 21:30
If i make a solution by adding 83grams of sodium hydroxide to 750ml i’d water what is the molarity of sodium hydroxide
Answers: 1

Chemistry, 22.06.2019 06:20
If i can still dissolve more sugar into the solution at a certain temperature what would i call that solution
Answers: 3

Chemistry, 22.06.2019 10:10
What shape would a molecule with two bound groups and two lone pairs have?
Answers: 1
You know the right answer?
Ammonia (nh3) boils at -33∘c; at this temperature it has a density of 0.81 g/cm3. the enthalpy of f...
Questions

Arts, 10.09.2020 03:01







English, 10.09.2020 03:01



History, 10.09.2020 03:01

Computers and Technology, 10.09.2020 03:01



French, 10.09.2020 03:01


Mathematics, 10.09.2020 03:01


Biology, 10.09.2020 03:01

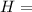 Δ
Δ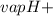 Δ
Δ