
Chemistry, 06.11.2019 02:31 kimbely7704
The molar heat capacity of ethane is represented in the temperature range 298 k to 400 k by the empirical expression cp, m in j k1 mol 14.73 + (0.1272 t in k). the corresponding expressions for c(e) and h2(g) are given in the back of the atkins textbook. calculate the standard enthalpy of formation of ethane at 373 k from its value at 298 k, in kj mol

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:30
Asa choose the correct set of reaction coefficients to properly balance the following chemical equation according to the law of conservation of mass: __s8 + __o2 ==> __so2 1, 1, 8 1, 8, 1 1, 8, 8 8, 1, 1
Answers: 1

Chemistry, 22.06.2019 07:00
6what is the importance of water on earth? a) it keeps the top layer of the geosphere cool b) it allows life to exist c) it provides ice at the poles d) it creates earth's blue color from space
Answers: 2

Chemistry, 22.06.2019 09:00
The nuclear fission process releases neutrons and question 27 options: alpha particles electrons energy beta particles
Answers: 1

Chemistry, 23.06.2019 01:00
Which is true concerning the products and reactants of photosynthesis and cellular respiration? a. the products of photosynthesis are sugars and the reactants of cellular respiration are starches. b. the products of photosynthesis are reactants in cellular respiration. c. oxygen is needed for photosynthesis and is given off in cellular respiration.
Answers: 2
You know the right answer?
The molar heat capacity of ethane is represented in the temperature range 298 k to 400 k by the empi...
Questions


Mathematics, 21.07.2019 13:40

Biology, 21.07.2019 13:40

History, 21.07.2019 13:40

History, 21.07.2019 13:40

History, 21.07.2019 13:40


Biology, 21.07.2019 13:40

Mathematics, 21.07.2019 13:40

History, 21.07.2019 13:40

Physics, 21.07.2019 13:40

Business, 21.07.2019 13:40


Biology, 21.07.2019 13:40


Physics, 21.07.2019 13:40




Spanish, 21.07.2019 13:40

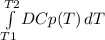
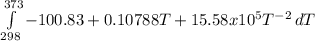 = -3796.48 J/mol = -3.80 kJ/mol (solved by a graphic calculator)
= -3796.48 J/mol = -3.80 kJ/mol (solved by a graphic calculator)

