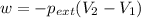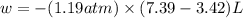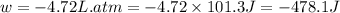Asample of gas in a cylinder of volume 3.42 l at 298 k and 2.57 atm expands to 7.39 l by two different pathways. path a is an isothermal, reversible expansion. path b has two steps. in the fi rst step, the gas is cooled at constant volume to 1.19 atm. in the second step, the gas is heated and allowed to expand against a constant external pressure of 1.19 atm until the fi nal volume is 7.39 l. calculate the work for each path.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 14:30
Which statement justifies that phosphine (ph3) is a polar molecule?
Answers: 1

Chemistry, 22.06.2019 10:00
Miner's coal distributors does not mine coal itself, nor does it even store or handle the coal. instead, miner's solicits orders for low sulfur coal from other firms, then purchases the required amount from suppliers and directs them to ship the coal to its customers. what is miner's
Answers: 1

Chemistry, 22.06.2019 10:20
Gwhich r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? which r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? −ch2−oh −ch2−o||c−nh2 −ch2−coo− −ch2−ch2−ch2−ch2−n+h3
Answers: 3

Chemistry, 22.06.2019 12:00
What is the percentage of hydrogen in nitrogen trihydride
Answers: 1
You know the right answer?
Asample of gas in a cylinder of volume 3.42 l at 298 k and 2.57 atm expands to 7.39 l by two differe...
Questions

Mathematics, 23.08.2020 02:01

Mathematics, 23.08.2020 02:01



Computers and Technology, 23.08.2020 02:01

Mathematics, 23.08.2020 02:01


Mathematics, 23.08.2020 02:01



History, 23.08.2020 02:01



Social Studies, 23.08.2020 02:01

Mathematics, 23.08.2020 02:01

Advanced Placement (AP), 23.08.2020 02:01


Mathematics, 23.08.2020 02:01


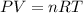
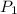 = initial pressure of gas = 2.57 atm
= initial pressure of gas = 2.57 atm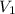 = initial volume of gas = 3.42 L
= initial volume of gas = 3.42 L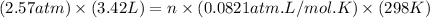
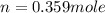
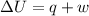
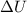 = internal energy
= internal energy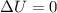
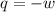
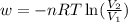
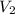 = final volume of gas = 7.39 L
= final volume of gas = 7.39 L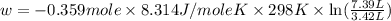
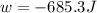
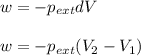
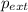 = external pressure = 1.19 atm
= external pressure = 1.19 atm