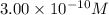Asolution contains 0.021 m cl and 0.017 m i. a solution containing copper (i) ions is added to selectively precipitate one of the ions. at what concentration of copper (i) ion will a precipitate begin to form? what is the identity of the precipitate? ksp(cucl) = 1.0 × 10-6, ksp(cui) = 5.1 × 10-12.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 21:30
The reaction q+r2=r2q is found to be first order in r2 and
Answers: 1

Chemistry, 21.06.2019 23:00
Plz choose one of the compounds from the table and explain how you know the numbers of atoms in your formula. is it possible for two different compounds to be made from the exact same two elements? why or why not? with a limited number of elements (less than 120 are known), does this mean we also have a small number of compounds or do we have a large number of compounds in this world?
Answers: 1

Chemistry, 22.06.2019 10:30
Which characteristics can be used to differentiate star systems? check all that apply.
Answers: 2

Chemistry, 22.06.2019 21:30
The solid xy decomposes into gaseous x and y: xy(s) m x(g) + y(g) kp = 4.1 (at 0 °c) if the reaction is carried out in a 22.4 l container, which initial amounts of x and y will result in the formation of solid xy?
Answers: 1
You know the right answer?
Asolution contains 0.021 m cl and 0.017 m i. a solution containing copper (i) ions is added to selec...
Questions


English, 04.12.2019 22:31




Biology, 04.12.2019 22:31

Computers and Technology, 04.12.2019 22:31

Mathematics, 04.12.2019 22:31







Mathematics, 04.12.2019 22:31

Mathematics, 04.12.2019 22:31




Mathematics, 04.12.2019 22:31

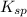 of CuCl =
of CuCl = 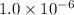
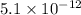
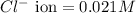
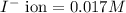
![K_{sp}=[Cu^+][Cl^-]](/tpl/images/0361/6888/73cc7.png)
![1.0\times 10^{-6}=[Cu^+]\times 0.021](/tpl/images/0361/6888/d87ff.png)
![[Cu^+]=\frac{1.0\times 10^{-6}}{0.021}=4.76\times 10^{-5}M](/tpl/images/0361/6888/3549a.png)
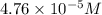
![K_{sp}=[Cu^+][I^-]](/tpl/images/0361/6888/38825.png)
![5.1\times 10^{-12}=[Cu^+]\times 0.017](/tpl/images/0361/6888/99563.png)
![[Cu^+]=\frac{5.1\times 10^{-12}}{0.017}=3.00\times 10^{-10}M](/tpl/images/0361/6888/762ae.png)