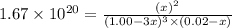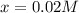Chemistry, 07.11.2019 06:31 winterblanco
0.0200 m fe3+ is initially mixed with 1.00 m oxalate ion, c2o42-, and they react according to the equation: fe3+(aq) + 3 c2o42-(aq) ⇄ [fe(c2o4)3]3-(aq) kc = 1.67 × 1020 what is the concentration of fe3+(aq) when equilibrium is reached? 1.67 × 1020 m 8.35 × 10-19 m 6.9a × 1021 m 1.44 × 10-22 m 0.980 am 0.940 m 0.0100 atm

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Asample of neon occupies a volume of 375 ml at stp. what will be the volume of neon if the pressure is reduced to 90.0 kpa? a. 422 ml b. 422 l c. 333 ml d. 333 l
Answers: 2

Chemistry, 22.06.2019 10:40
Ammonia and oxygen react to form nitrogen monoxide and water, like this: 4nh3 (g) + 5o2 (g) → 4no (g) + 6h2o (g) also, a chemist finds that at a certain temperature the equilibrium mixture of ammonia, oxygen, nitrogen monoxide, and water has the following composition: compound pressure at equilibrium nh3 65.1atm o2 31.3atm no 62.7atm h2o 65.8atm compound pressure at equilibrium nh3 65.3 atm o2 7.79 atm no 12.1 atm h2o 65.8 atm calculate the value of the equilibrium constant kp for this reaction. round your answer to 2 significant
Answers: 2


You know the right answer?
0.0200 m fe3+ is initially mixed with 1.00 m oxalate ion, c2o42-, and they react according to the eq...
Questions






Mathematics, 22.05.2020 08:02

English, 22.05.2020 08:02

Mathematics, 22.05.2020 08:02

Mathematics, 22.05.2020 08:02




Mathematics, 22.05.2020 08:02

Mathematics, 22.05.2020 08:02

Mathematics, 22.05.2020 08:02



Mathematics, 22.05.2020 08:02


Computers and Technology, 22.05.2020 08:02

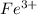 at equilibrium is 0 M.
at equilibrium is 0 M.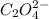 = 1.00 M
= 1.00 M![Fe^{3+}(aq)+3C_2O_4^{2-}(aq)\rightleftharpoons [Fe(C_2O_4)_3]^{3-}(aq)](/tpl/images/0363/4634/1f551.png)
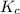 will be,
will be,![K_c=\frac{[[Fe(C_2O_4)_3]^{3-}]}{[C_2O_4^{2-}]^3[Fe^{3+}]}](/tpl/images/0363/4634/137fa.png)