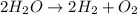Answers: 2


Another question on Chemistry



Chemistry, 22.06.2019 16:30
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2

Chemistry, 22.06.2019 22:40
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization.a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution.part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1
You know the right answer?
The following balanced equation shows the decomposition of water (h2o). according to the equation, h...
Questions



Mathematics, 23.08.2019 16:50

Mathematics, 23.08.2019 16:50


English, 23.08.2019 16:50


Mathematics, 23.08.2019 16:50

Biology, 23.08.2019 16:50


Mathematics, 23.08.2019 16:50

Advanced Placement (AP), 23.08.2019 16:50

Mathematics, 23.08.2019 16:50

History, 23.08.2019 16:50




History, 23.08.2019 16:50


Mathematics, 23.08.2019 16:50