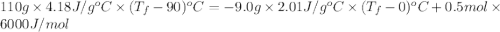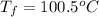Chemistry, 10.11.2019 03:31 julliette27
An ice cube of mass 9.0 g at temperature 0∘c is added to a cup of coffee, whose temperature is 90 ∘c and which contains 110 g of liquid. assume the specific heat capacity of the coffee is the same as that of water. the heat of fusion of ice (the heat associated with ice melting) is 6.0 kj/mol.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 10:00
Diffraction is when light is bent around obstructions. which of the these observation about clouds would indicate diffraction? a) after rain storms, you can sometimes see rainbows. b) clouds are white or gray and cannot be seen through. c) on a cloudy day, the temperature tends to be cooler than a sunny day. d) the edges of dark clouds appear lighter. this
Answers: 3

Chemistry, 22.06.2019 12:10
If a molecule with a molecular formula of c13h18 is treated with an excess of h2 in the presence of finally divided pt metal under conditions required for maximum hydrogenation of the molecule to give a molecule with a formula c13h24, how many rings are in the molecule?
Answers: 3

Chemistry, 22.06.2019 12:20
Consider the reaction of a(g) + b(g) + c(g) => d(g) for which the following data were obtained: experiment initial [a], mol/l initial [b], mol/l initial [c], mol/l initial rate, mol/l.s 1 0.0500 0.0500 0.0100 6.25 x 10^-3 2 0.100 0.0500 0.0100 2.50 x 10^-2 3 0.100 0.100 0.0100 1.00 x 10^-1 4 0.0500 0.0500 0.0200 6.25 x 10^-3 what is the rate law for the reaction?
Answers: 3

Chemistry, 22.06.2019 18:00
Mercury turns to vapor at 629.88 k how much heat is lost 175 g of mercury vapor at 650 current condenses to a liquid at 297 ca mercury turns to weber at 629.88 kelvin how much he is lost 175 g of mercury vapor and 650 coming condensers to liquidate 297 kevin
Answers: 2
You know the right answer?
An ice cube of mass 9.0 g at temperature 0∘c is added to a cup of coffee, whose temperature is 90 ∘c...
Questions

Mathematics, 26.02.2021 15:20

Mathematics, 26.02.2021 15:20

History, 26.02.2021 15:20

English, 26.02.2021 15:20

Mathematics, 26.02.2021 15:20

Mathematics, 26.02.2021 15:20

Chemistry, 26.02.2021 15:20

History, 26.02.2021 15:20





Mathematics, 26.02.2021 15:20

Physics, 26.02.2021 15:20

English, 26.02.2021 15:20

Computers and Technology, 26.02.2021 15:20




Chemistry, 26.02.2021 15:20

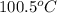
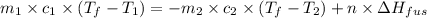
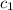 = specific heat of coffee = specific heat of water =
= specific heat of coffee = specific heat of water = 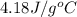 (as per question)
(as per question)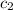 = specific heat of ice =
= specific heat of ice = 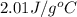
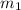 = mass of coffee = 110 g
= mass of coffee = 110 g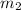 = mass of ice = 9.0 g
= mass of ice = 9.0 g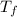 = final temperature = ?
= final temperature = ?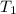 = initial temperature of coffee =
= initial temperature of coffee = 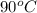
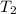 = initial temperature of ice =
= initial temperature of ice = 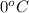
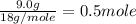
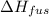 = 6.0 kJ/mol = 6000 J/mol
= 6.0 kJ/mol = 6000 J/mol