
Nh4no3, whose heat of solution is 25.7 kj/mol, is one substance that can be used in cold pack. if the goal is to decrease the temperature from 25.0 °c to 5.0 °c, how many grams of nh4no3 should we use for every 100.0 g of water in the cold pack? assume no heat was lost outside of cold pack, and the specific heat of the resulted solution was the same as water, or 4.184 j/(g•°c).

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:00
The alkali metals (group 1) consist of lithium (3), sodium (11), potassium (19), rubidium (37), cesium (55), and francium (87). they are soft, metallic solids with low densities and low melting points. based on the data shown in figure 1, how many valence electrons do alkali metals share?
Answers: 3

Chemistry, 22.06.2019 10:00
Select all of the methods through which a drug can enter your body. injection swallowing inhalation absorption
Answers: 2

Chemistry, 22.06.2019 14:30
Calculate the mass of carbon in 97.0 g of sucrose c12h22o11
Answers: 3

Chemistry, 22.06.2019 20:00
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3
You know the right answer?
Nh4no3, whose heat of solution is 25.7 kj/mol, is one substance that can be used in cold pack. if th...
Questions

Mathematics, 10.10.2019 11:00

Mathematics, 10.10.2019 11:00

Mathematics, 10.10.2019 11:00

Mathematics, 10.10.2019 11:10



Mathematics, 10.10.2019 11:10

Computers and Technology, 10.10.2019 11:10



Chemistry, 10.10.2019 11:10


Social Studies, 10.10.2019 11:10

History, 10.10.2019 11:10


Chemistry, 10.10.2019 11:10




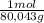 X
X

