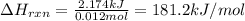Chemistry, 19.11.2019 00:31 keyonaemanieevans
Manganese reacts with hydrochloric acid to produce manganese(ii) chloride and hydrogen gas. mn(s)+2hcl(aq) ? mncl2(aq)+h2(g)
part a when 0.650g mn is combined with enough hydrochloric acid to make 100.0 ml of solution in a coffee-cup calorimeter, all of the mn reacts, raising the temperature of the solution from 23.0? c to 28.2? c. find ? hrxn for the reaction as written. (assume that the specific heat capacity of the solution is 4.18 j/g? c and the density is 1.00 g/ml.) express your answer using three significant figures. ? hrxn = kj

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 09:00
Which process does not require the presence of a physical substance in order to transfer heat? air in the atmosphere is heated by the ground. this warm air then rises, and cooler air falls. this is an example of what type of process? how is conduction different from radiation?
Answers: 1

Chemistry, 23.06.2019 05:30
Awhite powder is added to a solution. the images show observations made before the powder is added, just after the powder has been added, and a little while later. (the liquid in the small beaker is phenol red solution.) what evidence shows that a chemical change has taken place?
Answers: 1

Chemistry, 23.06.2019 07:30
Using this reversible reaction, answer the questions below: n2o4 2no2 (colorless) (reddish-brown) -as the temperature increased, what happened to the n2o4 concentration? -was the formation of reactants or products favored by the addition of heat? -which reaction is exothermic? right to left or left to right? -if the change of enthalpy of this reaction when proceeding left to right is 14 kcal, which chemical equation is correct? n2o4 2no2 + 14 kcal n2o4 2no2, hr = +14 kcal n2o4 + 14 kcal 2no2 n2o4 2no2, hr = -14 kcal
Answers: 1

Chemistry, 23.06.2019 10:00
The image shows the process of which is used in nuclear power plants. photo attached
Answers: 1
You know the right answer?
Manganese reacts with hydrochloric acid to produce manganese(ii) chloride and hydrogen gas. mn(s)+2h...
Questions

Social Studies, 10.11.2020 21:50

Mathematics, 10.11.2020 21:50

Geography, 10.11.2020 21:50





Mathematics, 10.11.2020 21:50


Chemistry, 10.11.2020 21:50

Advanced Placement (AP), 10.11.2020 21:50

Spanish, 10.11.2020 21:50

Health, 10.11.2020 21:50

English, 10.11.2020 21:50


Advanced Placement (AP), 10.11.2020 21:50

Mathematics, 10.11.2020 21:50



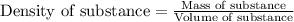
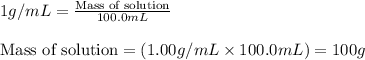
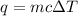
 = change in temperature =
= change in temperature = 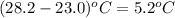
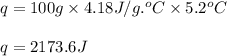
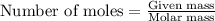
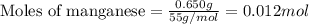
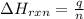
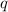 = amount of heat absorbed = 2173.6 J = 2.174 kJ (Conversion used: 1 kJ = 1000 J)
= amount of heat absorbed = 2173.6 J = 2.174 kJ (Conversion used: 1 kJ = 1000 J)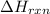 = enthalpy change of the reaction
= enthalpy change of the reaction