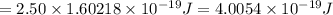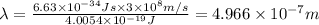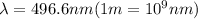Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 14:00
The two naturally occurring isotopes of chlorine are 35cl (34.969 amu, 75.77%) and 37cl (36.966 amu, 24.23%). the two naturally occurring isotopes of bromine are 79br (78.918 rm amu, 50.69%) and 81br (80.916 amu, 49.31%). chlorine and bromine combine to form bromine monochloride, brcl. 1. how many peaks will be present in a mass spectrum for brcl? the four combinations of molecule possible given these four isotopes are: 81br37cl, 81br35cl, 79br37cl, and 79br35cl. 2. what are the masses of the four different brcl molecules? express the masses using six significant figures, in decreasing numeric order (highest to lowest), separated by commas.
Answers: 3

Chemistry, 23.06.2019 01:00
What type of chemical bond is formed between two atoms of bromine 1. metallic 2. hydrogen 3. ionic 4. covalent
Answers: 1

Chemistry, 23.06.2019 02:00
Which best describes the present-day universe? opaque, expanding very slowly, stars produce heavy elements transparent, expanding at an accelerated rate, stars produce heavy elements opaque, expanding at an accelerated rate, stars produce only hydrogen and helium transparent, expanding very slowly, stars produce only hydrogen and helium
Answers: 1

Chemistry, 23.06.2019 07:30
Assignment directions: pick one of the following chemists and perform a bit of research on him/her. answer the following questions. alice hamilton rosalind franklin marie curie gertrude b. elion ada yonath henry cavendish robert boyle antoine lavoisier mario j. molina svante arrhenius
Answers: 1
You know the right answer?
For a specific molecule, the energy difference between the homo and lumo is 2.50 ev. what wavelength...
Questions

Chemistry, 14.02.2022 01:00

Chemistry, 14.02.2022 01:00

Mathematics, 14.02.2022 01:00


Biology, 14.02.2022 01:00



Mathematics, 14.02.2022 01:00

Mathematics, 14.02.2022 01:00

Social Studies, 14.02.2022 01:00

Mathematics, 14.02.2022 01:00


Mathematics, 14.02.2022 01:00

English, 14.02.2022 01:00


Social Studies, 14.02.2022 01:00

Biology, 14.02.2022 01:00

English, 14.02.2022 01:00


Mathematics, 14.02.2022 01:00

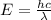
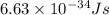
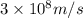
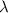 = wavelength = ?
= wavelength = ?