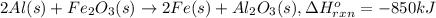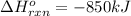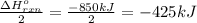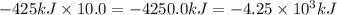Chemistry, 22.11.2019 03:31 lanashanabJHsbd1099
Calculate the enthalpy change for the thermite reaction: 2al(s)+fe2o3(s)→2fe(s)+al2o3(s), δh∘rxn=−850 kj when 10.0 mol of al undergoes the reaction with a stoichiometrically equivalent amount of fe2o3. express your answer to three significant figures and include the appropriate units.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 21:30
If i make a solution by adding 83grams of sodium hydroxide to 750ml i’d water what is the molarity of sodium hydroxide
Answers: 1

Chemistry, 22.06.2019 03:30
Asample of ammonia reacts with oxygen as shown. 4nh3(g) + 5o2(g) 4no(g) + 6h2o(g) what is the limiting reactant if 4.0 g of nh3 react with 8.0 g of oxygen? o2 because it produces only 0.20 mol of no. nh3 because it produces only 0.20 mol of no. o2 because it produces two times less no than nh3. nh3 because it produces three times more no than o2.
Answers: 3


Chemistry, 23.06.2019 09:00
Individuals within populations exhibit some diversity. as a result of possessing slightly different traits, some individuals are better able to survive and reproduce than others. if these individuals changes in the characteristics of the population may occur over time. the cumulative change in these characteristics is known as
Answers: 3
You know the right answer?
Calculate the enthalpy change for the thermite reaction: 2al(s)+fe2o3(s)→2fe(s)+al2o3(s), δh∘rxn=−8...
Questions



Biology, 28.08.2019 11:30


History, 28.08.2019 11:30

History, 28.08.2019 11:30


Mathematics, 28.08.2019 11:30

English, 28.08.2019 11:30

Mathematics, 28.08.2019 11:30

English, 28.08.2019 11:30

Social Studies, 28.08.2019 11:30



Health, 28.08.2019 11:30

Biology, 28.08.2019 11:30




 .
.